Density of water in gl2/9/2024  The most common unit is grams per milliliter (g/mL), which measures the mass of water in one milliliter volume.Īnother commonly used unit of measurement for water density is kilograms per cubic meter (kg/m3). 
When it comes to measuring water density, there are a variety of units of measurement that can be used. Remember that temperature also affects water density which we will be discussing later in this article.Īlso Read: Explore the Heart of Ivy League: Where is Harvard University located? The Different Units of Measurement Once you have obtained both measurements (mass and volume), simply divide them using the above formula to get your final result: water density in units such as grams per milliliter (g/mL) or kilograms per cubic meter (kg/m³). For example, you can use a graduated cylinder or pipette for smaller amounts of liquid or a volumetric flask for larger quantities. Be sure to tare the scale before measuring the weight of your container so that only the weight of the water is measured.įor measuring volume, there are different methods depending on your equipment and needs. To measure the mass of water, you can use a scale or balance. The formula for calculating water density is as follows: To calculate water density, you need to know the mass of water and its volume. Water density is an important factor in various industries and scientific studies. 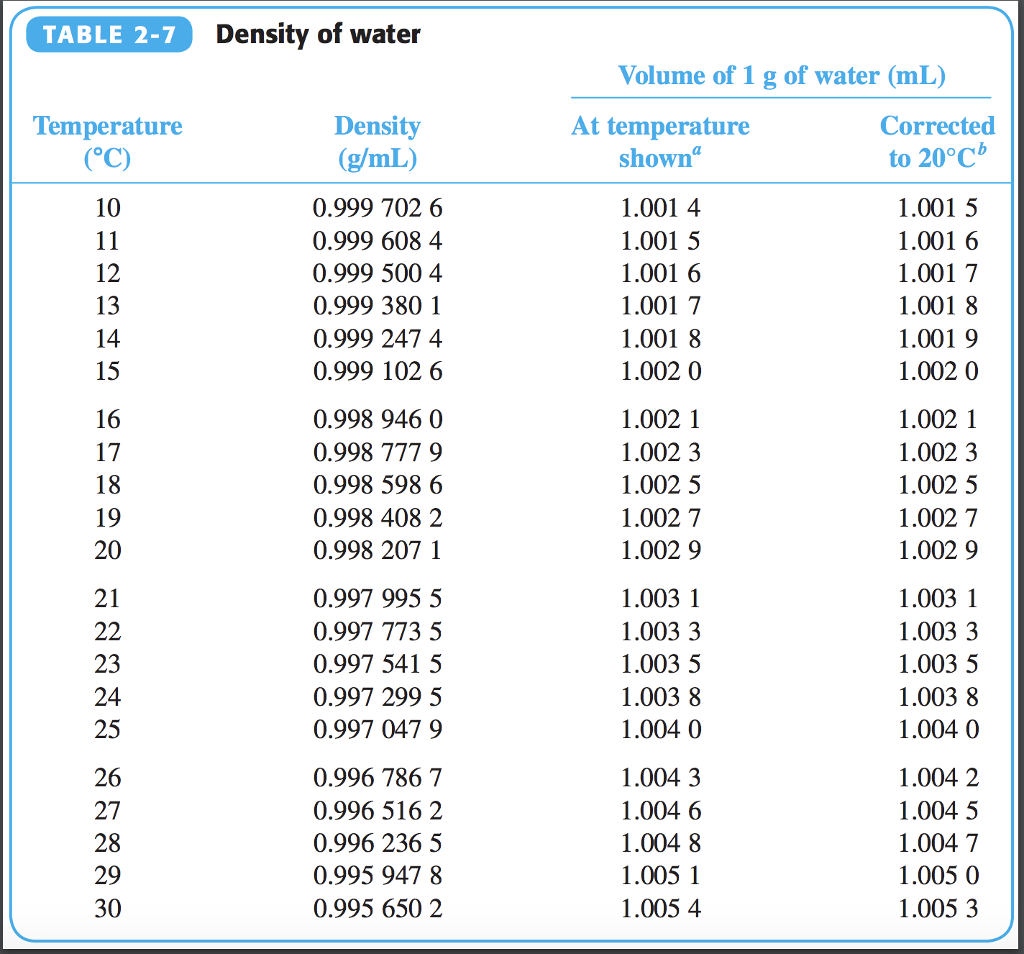
So grab your calculator and let’s dive into the fascinating world of water! How to Calculate Water Density Whether you’re a scientist or just curious about the properties of water, this guide will provide you with all the information you need to know about calculating water density. Understanding how to calculate water density at different temperatures and units is essential for various applications, such as designing pipelines, measuring liquid flow rates, and studying oceanography. Water density is a fundamental concept in physics and chemistry. 3 The Effect of Temperature on Water Density.Download Successful College Application Guide. 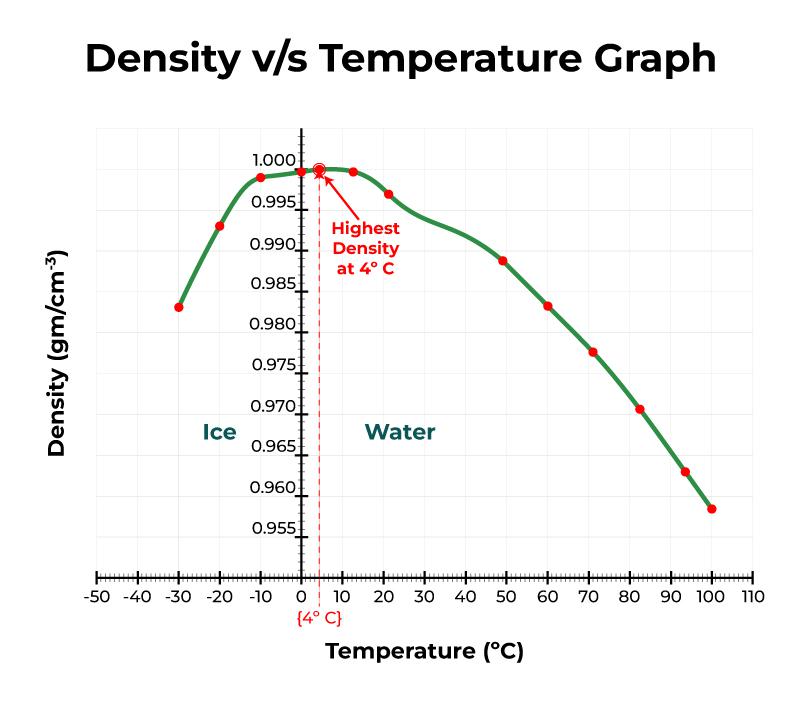
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
